The World Health Organization has expressed significant worry regarding the drug safety regulations in India following the report of a minimum of 20 children succumbing to tainted cough syrups.
The fatalities, occurring in Madhya Pradesh and Rajasthan regions within the past month, have been linked to three specific cough syrups. Examination of the products revealed the presence of a harmful substance known as diethylene glycol (DEG), commonly found in industrial solvents.
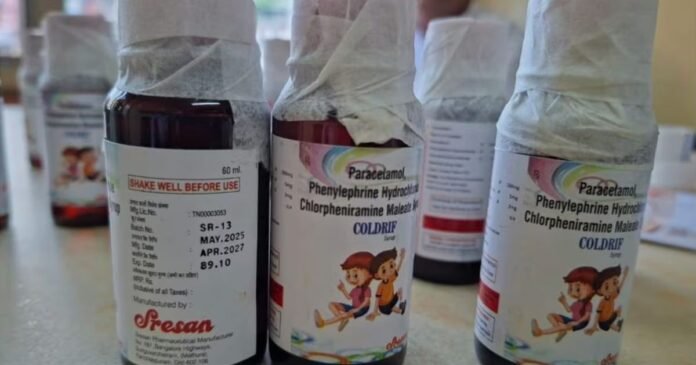
Identified by India’s drug regulator, the three contaminated syrups are Coldrif (Sresan Pharmaceuticals), Respifresh (Rednex Pharmaceuticals), and ReLife (Shape Pharma). Production of these medications has been halted, and an inquiry has been initiated.
Despite this action, the WHO has issued a caution that these cough syrups may enter other countries through unregulated distribution channels, as reported by the BBC. DEG is a toxic compound that can have fatal consequences even in small doses, especially for children, according to the WHO.
The organization emphasized that diethylene glycol (DEG) and ethylene glycol (EG) are typically utilized in industrial products like antifreeze and adhesives and should never be ingested by humans.
On Thursday, G Ranganathan, the 73-year-old owner of Sresan Pharmaceuticals, was apprehended. Tamil Nadu Health Minister Ma Subramaniam declared that the firm’s manufacturing license would be permanently revoked.
A scrutiny by the drug department revealed that Sresan Pharmaceuticals had breached 364 manufacturing regulations. The inspection report, highlighted by the BBC, revealed severe violations including unhygienic storage of manufactured products, improper sewage discharge, and inadequate water storage for drug production.
Additionally, the report noted deficiencies in production monitoring procedures, substandard water and equipment, and the absence of a data collection department.
This incident is not the first time concerns have been raised over Indian-manufactured cough syrups. In 2022, over 70 children in Gambia died from acute kidney failure after consuming imported cough syrup. Similarly, between 2022 and 2023, 68 children in Uzbekistan perished after ingesting another contaminated syrup from India.
India ranks as the third-largest drug producer globally by volume, following the United States and China. Pharmaceutical products are typically screened for DEG using gas chromatography. However, many laboratories in remote and under-resourced areas lack access to this technology, as noted by the WHO.
Earlier this year, the WHO initiated a public consultation on screening devices to detect DEG-contaminated medications. The organization highlighted the infiltration of DEG into pediatric syrups, resulting in numerous deaths across various countries.
The WHO stressed that DEG and EG can lead to multiorgan failure, particularly acute renal failure and neurological issues, causing numerous fatalities, particularly among children.